 
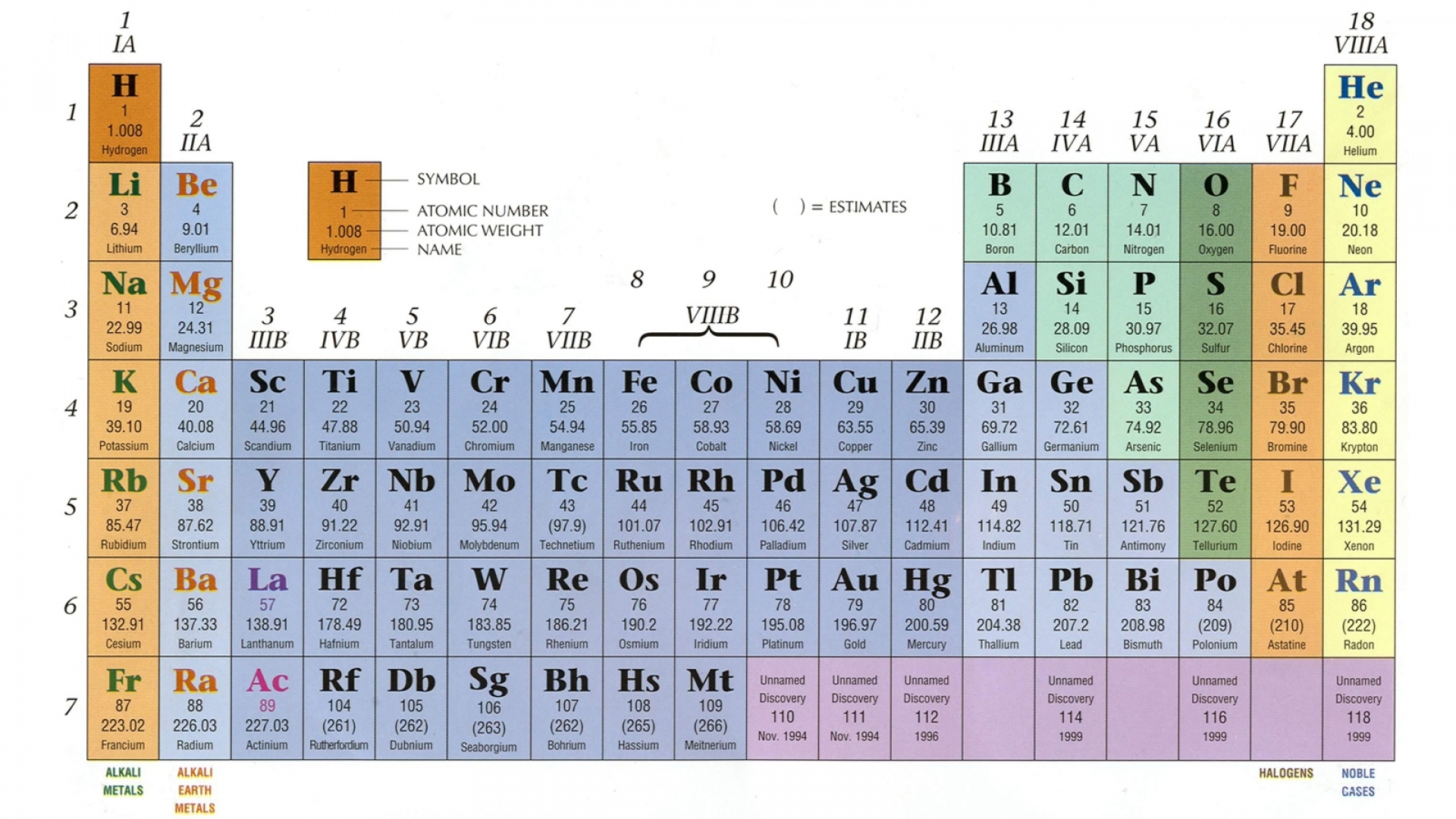
Hydrogen is a unique type of chemical element that does not found free in the earth’s atmosphere but is widely used in everyday life. The Greek words hydro and genes, meaning water forming These isotopes are used as an alternative fuel for engines or renewable energy sources for our environment. 
These three isotopes form three covalent gas molecules like dihydrogen, dideuterium, and ditritium. Hydrogen has the three isotopes having the chemical name protium, deuterium, and tritium and chemical formula 1H 1, 1H 2, and 1H 3 respectively. The most common oxidation number of hydrogen = +1, but due to unique characteristics, it also shows a −1 oxidation state to form LiH, NaH, CsH, and CaH 2. Due to the presence of the half-filled valence electrons, hydrogen is also placed in group-14 with the carbon family by forming a wide number of covalent chemical bonds with a wide range of polarity.All these elements have very high electronegativity and electron affinity. Therefore, it may be placed in group 17 with the halogen family such as fluorine, chlorine, bromine, and iodine. If we consider the electron configuration of hydrogen, it is just one electron short of the next noble gas helium.The ionization energy of hydrogen is also similar to that of alkali metals. The ns 1 electronic configuration hydrogen justifies the position in period-1 and group-1 with the alkali metals family ( lithium, sodium, potassium, rubidium, cesium, and francium).It is the third most abundant chemical element after oxygen and silicon and 2nd most abundant gas molecule after oxygen. The name hydrogen was given from the origin of Greek words meaning ‘water maker’.īesides the common physical state of matter like solid, liquid, and gas on the earth’s universe, hydrogen is a gas molecule at room temperature. In 1766 Henry Cavendish, an English chemist and physicist discovered the most combustible element or molecule like hydrogen. Under ordinary conditions, due to the presence of one valence electron in the 1s- orbital, it forms a wide number of common chemical bonding such as covalent, ionic, bridge bonds, and hydrogen bonding. It contains one electron and one proton with atomic number = 1 and atomic weight or mass = 1.007829. Atomic hydrogen has a very simple structural model. Approximately 100,000 gallons of water will produce a single gallon of D 2O, "heavy water".Hydrogen has the three isotopes having the chemical name protium ( 1H 1), deuterium ( 1H 2), and tritium ( 1H 3). Commonly indicated with symbol D and sometimes called heavy hydrogen, deuterium is separated by the fractional distillation of liquid hydrogen but it can also be produced by the prolonged electrolysis of ordinary water. Deuterium ( 2H ) is another an isotope containing a proton and neutron, consisting of only 0.0156% of the naturally occurring hydrogen. 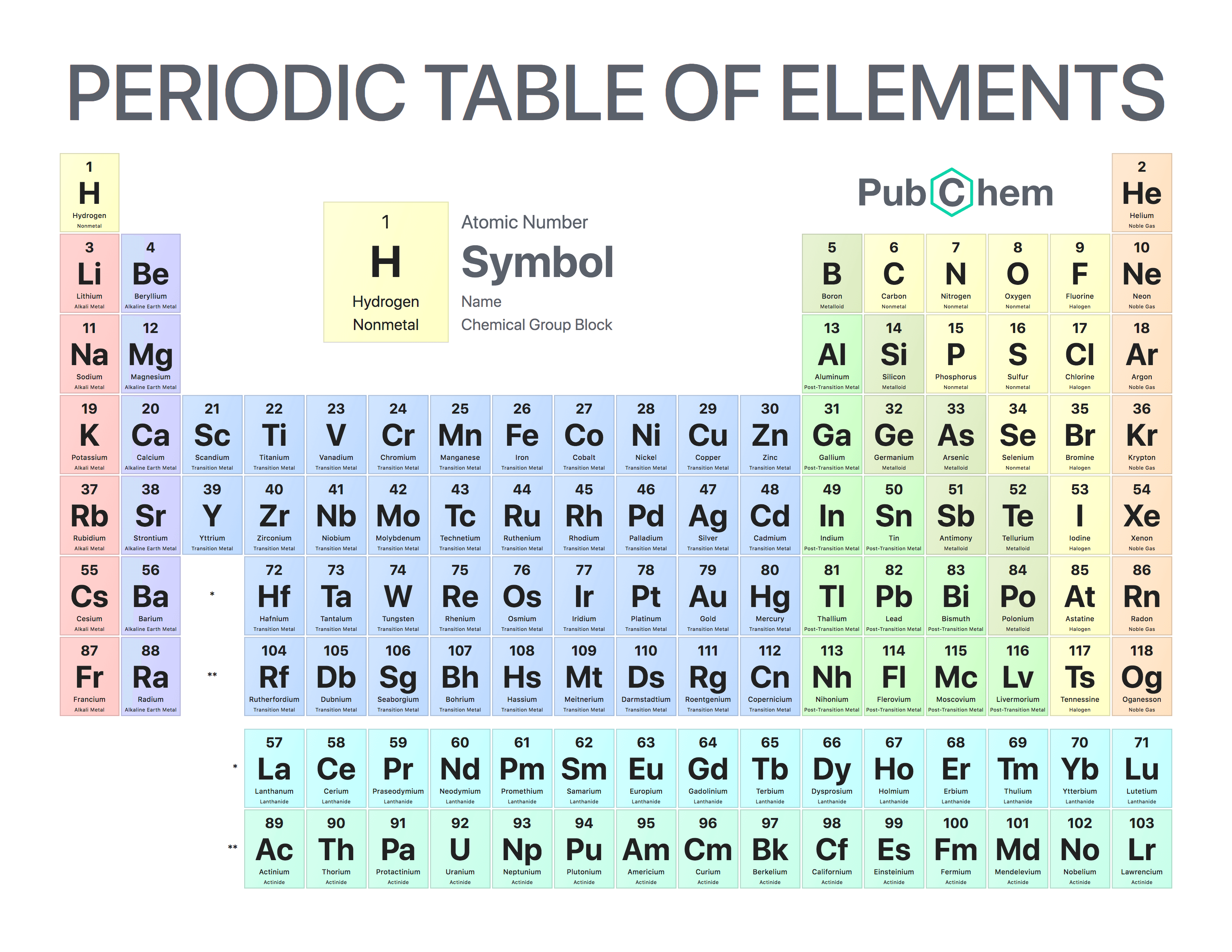
It is a nucleus containing a single proton.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
